Recent headlines suggested that the promising multi-cancer blood test developed by GRAIL had effectively failed. Much of the media coverage followed a narrative that the flagship screening test, Galleri, did not meet expectations in a large clinical trial. Stock prices dropped and the tone of many reports implied the technology might be finished.
However, a deeper analysis suggests the situation is far more nuanced. Physician and longevity researcher Peter Attia argues that the early conclusions circulating in the media may be premature.
Understanding the Galleri Test
Galleri is designed to detect multiple cancers from a single blood draw. The technology analyzes fragments of cell-free DNA circulating in the bloodstream. These are genetic material shed by tumors. Using a technique called targeted methylation sequencing, the test evaluates chemical modification patterns in DNA that differ between healthy tissue and cancerous tissue.
Rather than identifying a single mutation, Galleri looks for patterns associated with more than 50 different cancer types. When the test detects a cancer signal, it can also predict the likely organ of origin. This helps physicians focus diagnostic follow-up on the most probable location.
The test has been commercially available in the United States since 2021. It typically costs around $949 out-of-pocket because it is not yet widely covered by insurance.
The NHS-Galleri Trial
To evaluate whether the test could improve cancer detection, a massive study called the NHS-Galleri trial was launched through the National Health Service. The study included approximately 142,000 adults between the ages of 50 and 77. Participants were randomly assigned to either receive annual Galleri screening or continue with standard cancer screening protocols.
Ideally, cancer screening studies measure whether a test reduces cancer deaths. But mortality studies require extremely large populations and many years of follow-up. To make the trial more feasible, researchers used a proxy endpoint instead: whether the test reduced the number of late-stage cancers (Stage III and IV) diagnosed in the population.
When preliminary results were released, the trial did not meet its primary composite endpoint. This means the combined reduction of Stage III and Stage IV cancers was not statistically significant. This finding generated the dramatic headlines.
What the Headlines Missed
According to the analysis by Dr. Attia, the situation is more complicated than the headlines suggest. First, the data released so far came through a press release rather than a full peer-reviewed publication. Detailed results have not yet been publicly presented. The full dataset is expected to be discussed at a major oncology conference later this year.
Second, even within the limited information available, there were signals that could be clinically meaningful. In a predefined group of particularly deadly cancers—including pancreatic, liver, ovarian, lung, and stomach cancers—the trial appears to show a reduction in Stage IV diagnoses over repeated screening rounds.
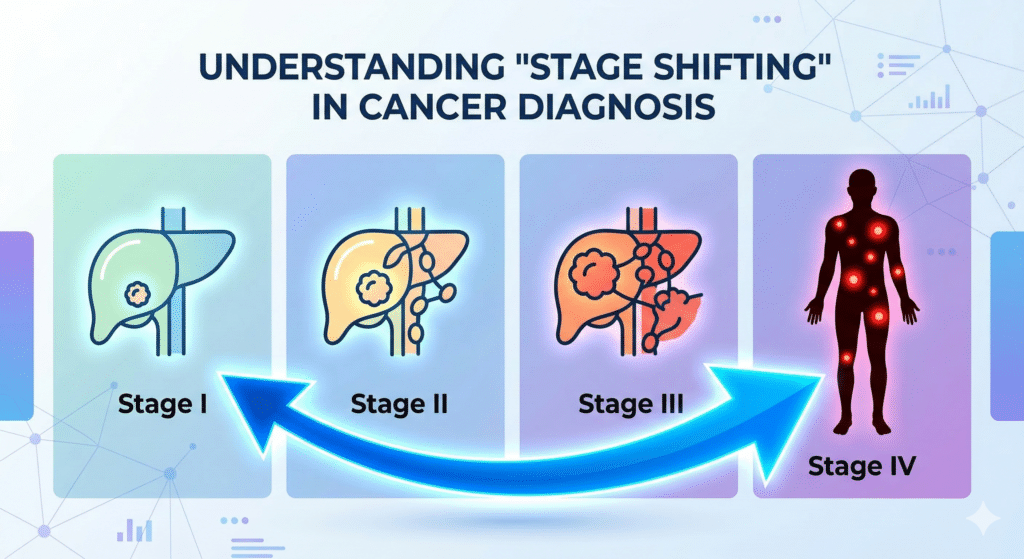
According to the company’s announcement, Stage IV diagnoses in these cancers declined by more than 20 percent in later screening rounds. At the same time, early-stage cancers (Stage I and II) were detected at higher rates compared with standard screening alone. This suggests the test may be shifting diagnoses earlier in the disease process. This is an outcome that could potentially improve survival, although longer follow-up will be needed to confirm that.
The Broader Screening Debate
The mixed interpretation of these results reflects a broader debate within medicine about cancer screening. Skeptics argue that screening tests can cause harm through false positives, unnecessary procedures, and overdiagnosis. For population-level guidelines, many experts insist on clear evidence that a test reduces mortality before recommending widespread adoption.
Others argue that the calculus may be different for individuals. For cancers such as pancreatic, ovarian, or liver cancer where there are currently no routine screening options, detecting even a small percentage of cases earlier could represent a meaningful improvement.
Why the Story Isn’t Finished
Galleri represents only one generation of multi-cancer early detection technology. Diagnostic platforms often evolve rapidly, improving in sensitivity and specificity over time. Early mammography technology was far less effective than modern imaging systems. Yet those early versions helped lay the groundwork for the screening programs widely used today.
The larger challenge facing the entire field is proving mortality reduction. Randomized trials capable of definitively answering that question would likely require hundreds of thousands of participants and more than a decade of follow-up. This makes them extraordinarily expensive and difficult to conduct. Because of this, researchers often rely on proxy measures such as stage shift, particularly reductions in Stage IV cancers.
A Balanced Perspective
At this stage, the most responsible interpretation may be the simplest: the Galleri trial did not deliver a clear victory, but it also did not prove the technology ineffective. The full data from the NHS-Galleri trial have not yet been published or peer-reviewed. Until that information is available, declaring the technology a failure or a success would be premature.
As Dr. Attia notes, the appropriate response is careful evaluation rather than sensational conclusions. The future of multi-cancer early detection will likely depend on continued research, improved technology, and thoughtful interpretation of emerging evidence. For now, the debate remains open and the potential impact of blood-based cancer screening continues to be one of the most important questions in modern preventive medicine.